Research
P2Network provides a platform for aspiring POCUS researchers to develop and refine research ideas, advancing POCUS knowledge by conferring and deciding on topics worthwhile of investment in research resources. Research projects are often multi-center, allowing researchers to collaborate and pool resources to investigate topics of common interests. We provide general mentorship, research protocol feedback and research mentorship.
P2 Network TOP TEN Articles of 2022
Ongoing Research
[email protected]
P2-Sponsored Projects
P2-sponsored projects receive general mentorship, research protocol feedback and research mentorship.
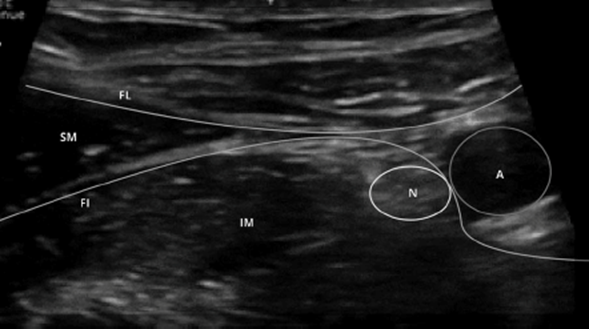
PLEXUS (Pediatric Lower Extremity Ultrasound-Guided Nerve Block) Study
P.I.: Zack Binder (U. of Mass)
Study Objectives: To compare ultrasound-guided fascia iliaca compartment nerve block (FICNB) to systemic analgesia in the treatment of pain from femur fractures
Current Status: IRB approved. Sites actively enrolling subjects. Multiple centers in IRB approval process. Recruiting other sites.
Email if interested: [email protected]
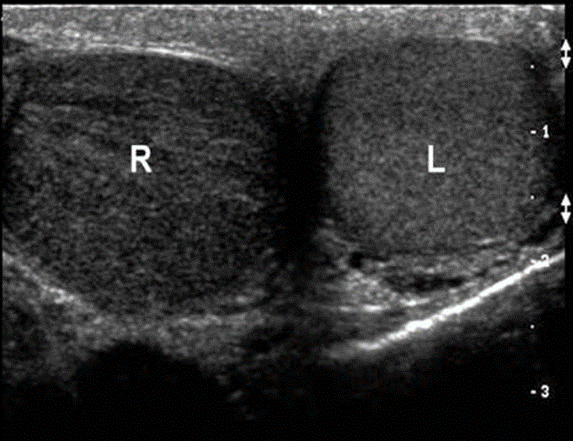
Investigating Scrotal POCUS in Pediatric Emergency Medicine
P.I.: Paul Khalil & Ee Tay (Nicklaus Children’s Hospital, NYU)
Study Objective: To investigate scrotal point-of-care ultrasound use in pediatric emergency medicine.
Current Status: First part of the ladder study is complete and document submitted. Second part of the retrospective study coming soon. Looking for a lead author for the prospective study.
Email if interested: [email protected], [email protected]
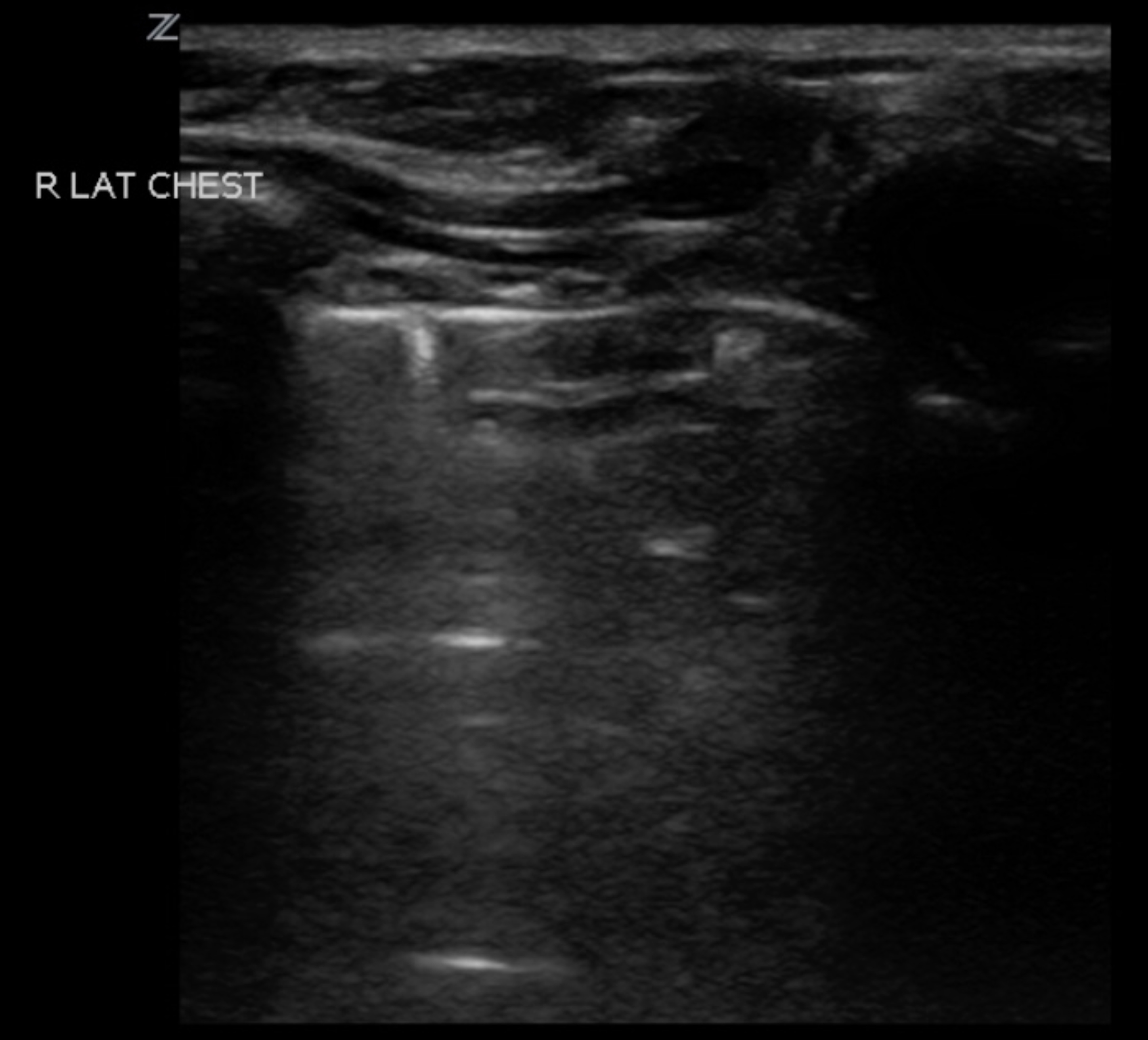
POCUS for Pediatric Thoracic Trauma: A Multi-Institutional Trial
P.I.: Kirstin Weerdenburg & Sam Lam (Sutter Medical Center Sacramento)
Study Objective: To determine the diagnostic accuracy of ED POCUS in pediatric traumatic thoracic injuries compared to chest x-ray/chest CT.
Current Status: Approved by Sutter IRB. Pilot sites have been selected and IRB submission begun. Internal grant awarded (Sutter).
Non-P2 Sponsored Projects
Independent projects seeking multi-site collaboration are listed below.

Teaching Select POCUS Applications to Pediatric Residents
P.I.: Marjan Z Ghazi Askar (Stanford)
Study Objectives:To design a curriculum to implement POCUS education amongst pediatric residency programs.
Current Status: Pilot survey complete, favoring pediatric residents seeking POCUS training during residency. POCUS subcommittee within the learning community of APPD created. Recruiting POCUS faculty to help design a curriculum to implement POCUS education amongst pediatric residency programs.
E-mail: If interested, email Marjan at [email protected]
P2 RESEARCH MENTORS
| NAME | CONTACT | INTERESTS | SPECIFIC INTERESTS |
| Kelly Bergmann | [email protected] | Educational Clinical Application Administrative |
|
| David Kessler | [email protected] | Educational Clinical Application |
AI/Machine learning Novel applications Lumbar puncture/procedures Augmented reality Simulation |
| Adib Salim | [email protected] | Educational Clinical Application |
|
| Ben Nti | [email protected] | Clinical Application | |
| Allan Shefrin | [email protected] | Educational Administrative |
Curriculum development Consensus building Delphi experience |
| Munaza Rizvi | [email protected] | Clinical Application | |
| Meshari Dalbouh | [email protected] | Clinical Application | Lung & Ocular POCUS |
| Paul Khalil | [email protected] | Educational Clinical Application Administrative |
|
| Adam Sivitz | [email protected] | Clinical Application | |
| Matthew Moake | [email protected] | Educational Clinical Application |
|
| Matthew Kusulas | [email protected] | Educational | |
| Alexander Arroyo | [email protected] | Educational Clinical Application Administrative |
Recent P2-Associated and Sponsored Publications
Snelling et al. Establishing the international Research Priorities for Pediatric Emergency Medicine Point-of-Care Ultrasound: A modified Delphi Study. Acad Emerg Med. Nov 2022.
https://pubmed.ncbi.nlm.nih.gov/36043227/
Bergmann et al. Accuracy and Interrater Reliability of Point-of-Care Ultrasonography Image Interpretation for Intussusception. Pediatric Emerg Care. Sep 2022.
https://pubmed.ncbi.nlm.nih.gov/36040465/
Kornblith et al. Development of a Consensus-Based Definition of Focused Assessment with Sonography for Trauma in Children. JAMA Netw Open. Mar 2022.
https://pubmed.ncbi.nlm.nih.gov/35302632/
Lam et al. The P2Network-Advancing Pediatric Emergency Care with Point-of-Care Ultrasound. Pediatric Emerg Care. Feb 2022.
https://pubmed.ncbi.nlm.nih.gov/34787985/
Bergmann et al. Diagnostic Accuracy of Point-of-Care Ultrasound for Intussusception: A Multi-center, Noninferiority Study of Paired Diagnostic Tests. Ann Emerg Med. Nov 2021.
https://pubmed.ncbi.nlm.nih.gov/34226072/
Lee et al. Variability in Point-of-Care Lung Ultrasound Findings in Pediatric COVID-19 Patients: A Multicenter Case Series. Pediatric Emerg Care. Dec 2021.
https://pubmed.ncbi.nlm.nih.gov/34772878/
Gold D et al. Design of a Point-of-Care Ultrasound Curriculum for Pediatric Emergency Medicine Fellows: A Delphi Study. AEM Educ Train. Aug 2021.
https://pubmed.ncbi.nlm.nih.gov/34901685/
Shefrin AE. Consensus Core Point-of-Care Ultrasound Applications for Pediatric Emergency Medicine Training. AEM Educ Train. Mar 2019.
https://pubmed.ncbi.nlm.nih.gov/31360818/
